(b): RNA and DNA molecules have ribose and deoxyribose sugar respectively. Both are chiral and their chirality is due to D-ribose or D-2-deoxyribose sugar component.
In DNA molecule, the sugar moiety is β-D-2-deoxyribose whereas in RNA molecule, it is β-D-ribose
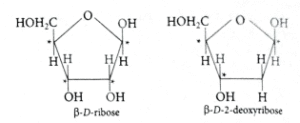
(b): RNA and DNA molecules have ribose and deoxyribose sugar respectively. Both are chiral and their chirality is due to D-ribose or D-2-deoxyribose sugar component.
In DNA molecule, the sugar moiety is β-D-2-deoxyribose whereas in RNA molecule, it is β-D-ribose

(b): RNA and DNA molecules have ribose and deoxyribose sugar respectively. Both are chiral and their chirality is due to D-ribose or D-2-deoxyribose sugar component.
In DNA molecule, the sugar moiety is β-D-2-deoxyribose whereas in RNA molecule, it is β-D-ribose
