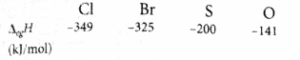4. Given below are two statements:
Statement I: The correct order in terms of atomic/ ionic radii is Al > Mg > Mg^2+ > Al^3+.
Statement II: The correct order in terms of the magnitude of electron gain enthalpy is Cl > Br > S > O.
In the light of the above statements, choose the correct answer from the options given below:
(d): Atomic radii decreases on moving from left to right in a period hence, atomic radius of Mg is greater than Al while among isoelectronic species radii decreases with increase in positive charge. This happens because effective as nuclear change (Z_eff) increases.
Hence, the final order id Mg > Al > mg^2+>Al^3+
Values of electron gain enthalpies are given as.
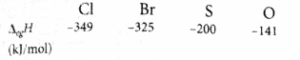
(d): Atomic radii decreases on moving from left to right in a period hence, atomic radius of Mg is greater than Al while among isoelectronic species radii decreases with increase in positive charge. This happens because effective as nuclear change (Z_eff) increases.
Hence, the final order id Mg > Al > mg^2+>Al^3+
Values of electron gain enthalpies are given as.
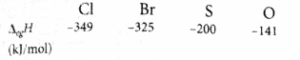
(d): Atomic radii decreases on moving from left to right in a period hence, atomic radius of Mg is greater than Al while among isoelectronic species radii decreases with increase in positive charge. This happens because effective as nuclear change (Z_eff) increases.
Hence, the final order id Mg > Al > mg^2+>Al^3+
Values of electron gain enthalpies are given as.